Aqueous Crystal Growth and Dissolution Kinetics of Earth Surface Materials
Although calcium carbonate reaction kinetics has important application in several areas of Earth Science, the mechanism of natural organic matter mediation of carbonate minerals growth and dissolution rates remains largely unknown. This project uses multiple approaches to study calcium carbonate formation and dissolution rates in surface water and groundwater systems.
Background
Project work on calcium carbonate reaction kinetics has important application in several areas of earth science, including water quality concerns and the global carbon budget. The mechanism of natural organic matter mediation of carbonate minerals growth and dissolution rates remains largely unknown. We use laboratory, field, and theoretical approaches to study calcium carbonate formation and dissolution rates in surface water and groundwater systems.
Important aspects of project research include characterization of the interaction of natural organic material with calcium carbonate surfaces during crystal growth and dissolution. We develop models to evaluate natural organic material influence on calcium carbonate crystal growth and dissolution kinetics. Other project activities focus on the development and verification of methods to characterize natural organic matter-metal ion speciation.
Project staff sample surface and ground water for chemical and isotopic composition related to calcium carbonate formation and dissolution. We use geochemical mass balance calculations to identify possible calcium carbonate formation and dissolution reactions in a range of surface and ground water settings. Project staff also monitor calcium carbonate crystal growth and dissolution in conjunction with tools developed for dating young groundwaters.
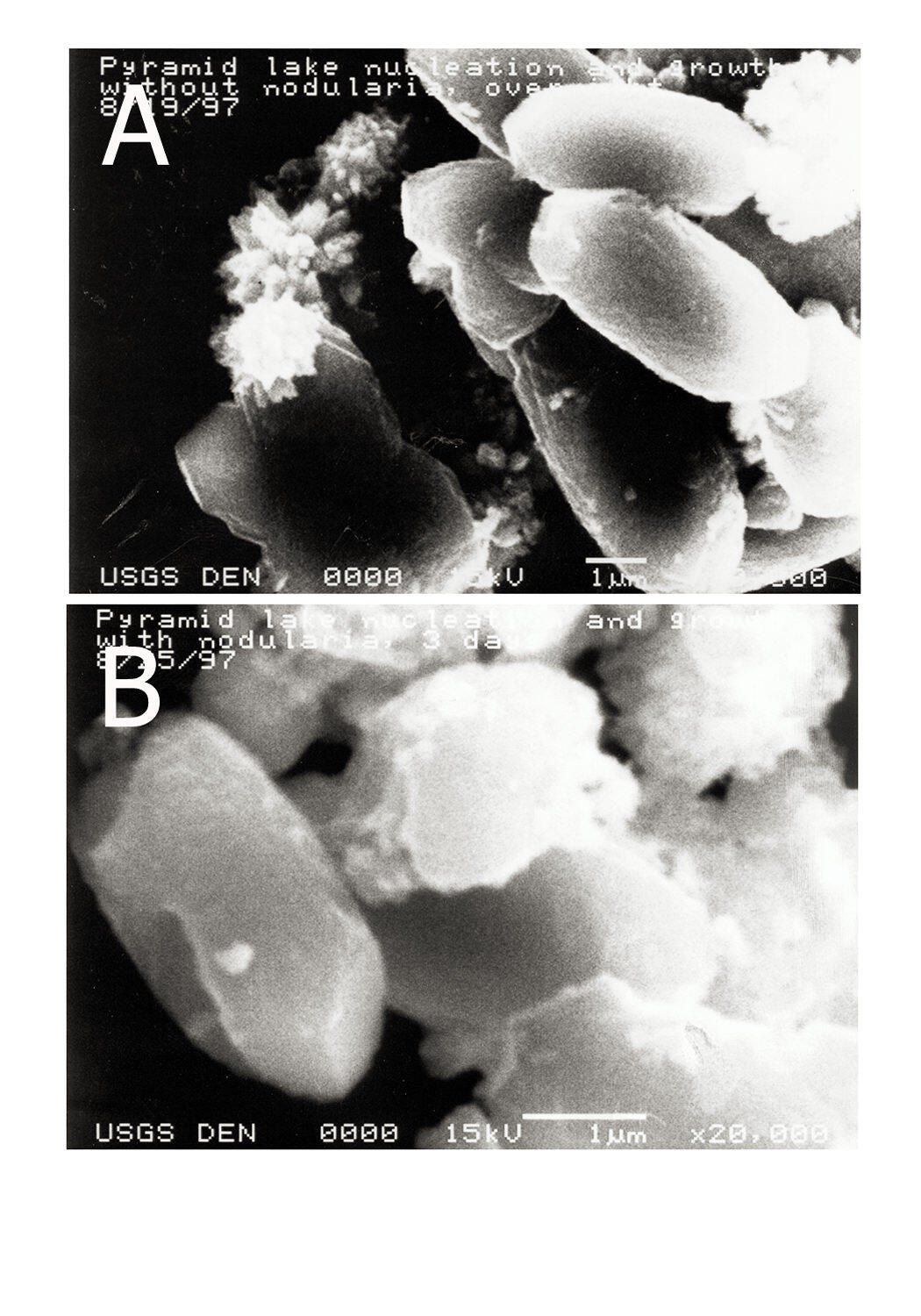
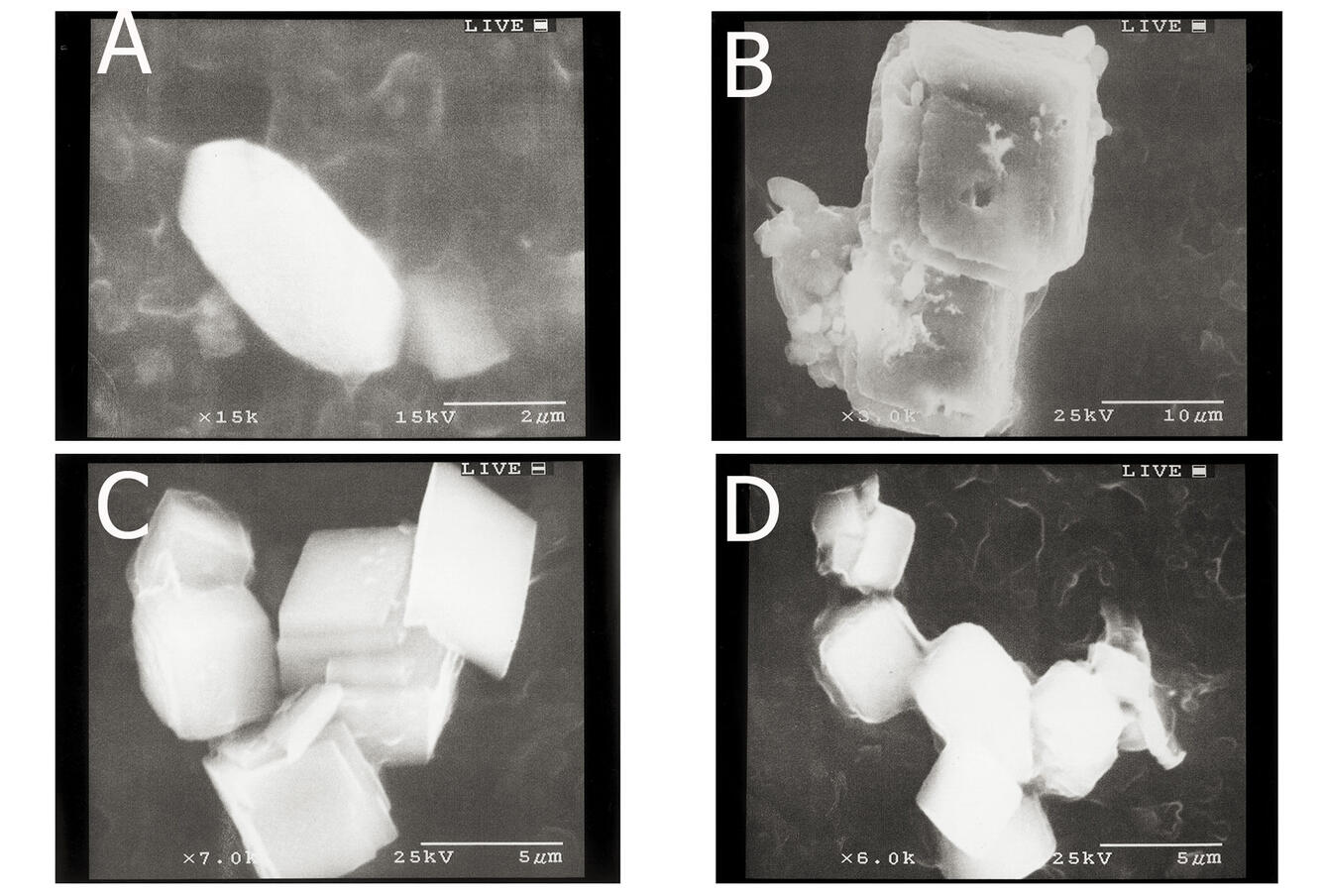
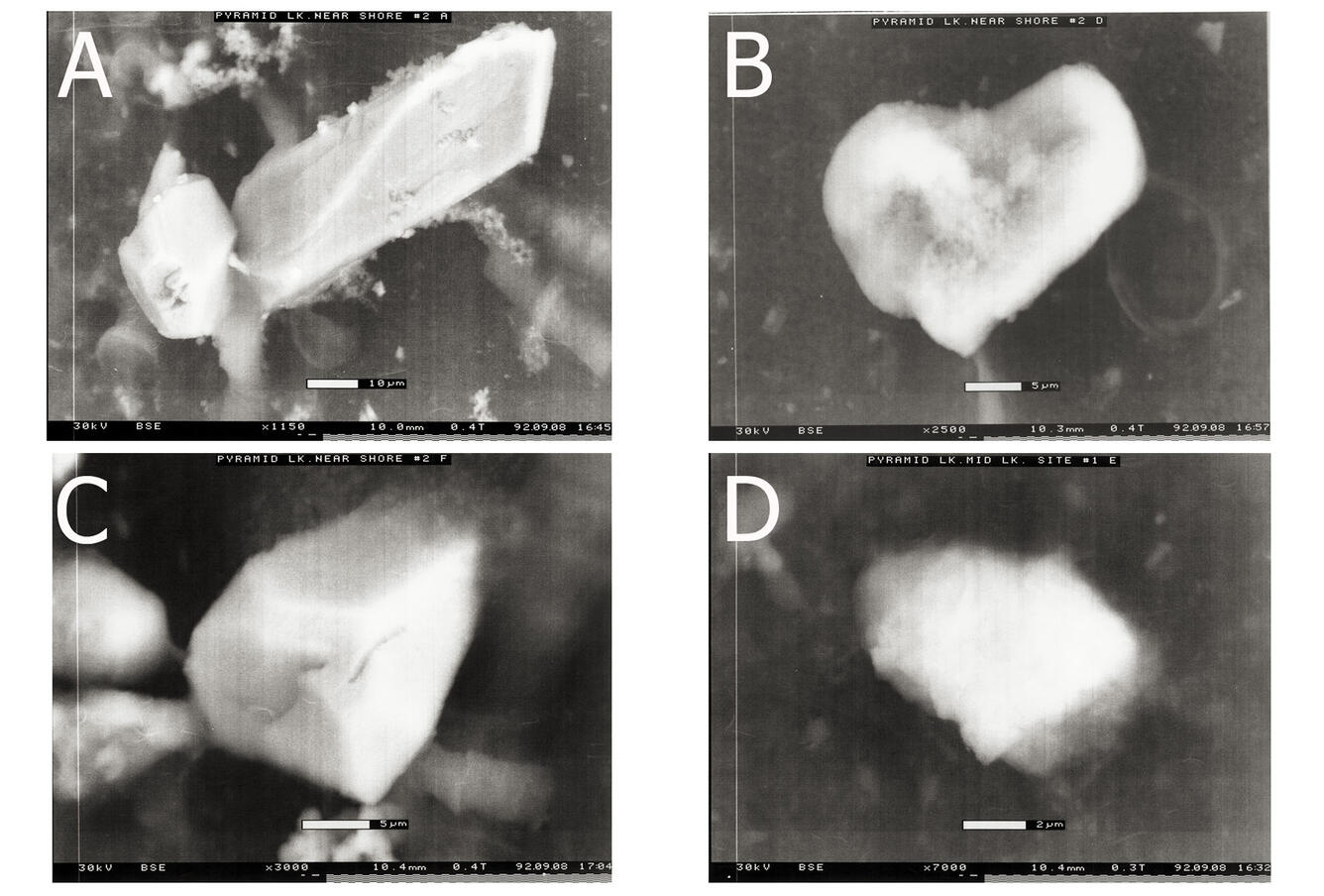
Our project uses techniques of low-temperature geochemistry to interpret and characterize chemical processes, such as precipitation of calcium carbonate in surface lake water (see photos below), occurring in surface water and ground water. We study metal binding by dissolved organic matter and crystal growth and dissolution reaction rate mediation by dissolved constituents because metal ion speciation and mineral formation rates influence water quality and carbon storage. Metal ion complexation with organic matter mediates metal ion reactivity in surface water and ground water -- metal ion speciation controls metal ion chemical and biogeochemical reactivity and bioavailability.
We determine equilibrium metal species distributions in surface and ground water containing organic matter, and characterize crystal growth and dissolution rates in surface and ground water containing organic material.
Project Interests
- Calcium carbonate nucleation and growth in surface and groundwater
- Calcium carbonate formation and dissolution in arid soils at the Amargosa Desert Research Site located near Beatty, Nevada.
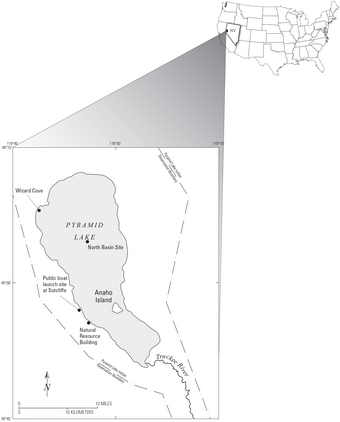
Pyramid Lake Research



Aqueous Crystal Growth and Dissolution Kinetics of Calcium Carbonate Minerals at the Amarigosa Desert Research Site, Nevada
Below are publications associated with this project.
Specific surface area of a crushed welded tuff before and after aqueous dissolution
Effects of acid rain and sulfur dioxide on marble dissolution
Welded tuff porosity characterization using mercury intrusion, nitrogen and ethylene glycol monoethyl ether sorption and epifluorescence microscopy
Employment of Gibbs-Donnan-based concepts for interpretation of the properties of linear polyelectrolyte solutions
Modeling electrostatic and heterogeneity effects on proton dissociation from humic substances
Description and testing of three moisture sensors for measuring surface wetness on carbonate building stones
Interpretation with a Donnan-based concept of the influence of simple salt concentration on the apparent binding of divalent ions to the polyelectrolytes polystyrenesulfonate and dextran sulfate
Proton and metal ion binding to natural organic polyelectrolytes-I. Studies with synthetic model compounds
Proton and metal ion binding to natural organic polyelectrolytes-II. Preliminary investigation with a peat and a humic acid
Crystal growth of calcite from calcium bicarbonate solutions at constant PCO2 and 25°C: a test of a calcite dissolution model
Below are software products associated with this project.
Although calcium carbonate reaction kinetics has important application in several areas of Earth Science, the mechanism of natural organic matter mediation of carbonate minerals growth and dissolution rates remains largely unknown. This project uses multiple approaches to study calcium carbonate formation and dissolution rates in surface water and groundwater systems.
Background

Project work on calcium carbonate reaction kinetics has important application in several areas of earth science, including water quality concerns and the global carbon budget. The mechanism of natural organic matter mediation of carbonate minerals growth and dissolution rates remains largely unknown. We use laboratory, field, and theoretical approaches to study calcium carbonate formation and dissolution rates in surface water and groundwater systems.
Important aspects of project research include characterization of the interaction of natural organic material with calcium carbonate surfaces during crystal growth and dissolution. We develop models to evaluate natural organic material influence on calcium carbonate crystal growth and dissolution kinetics. Other project activities focus on the development and verification of methods to characterize natural organic matter-metal ion speciation.
Project staff sample surface and ground water for chemical and isotopic composition related to calcium carbonate formation and dissolution. We use geochemical mass balance calculations to identify possible calcium carbonate formation and dissolution reactions in a range of surface and ground water settings. Project staff also monitor calcium carbonate crystal growth and dissolution in conjunction with tools developed for dating young groundwaters.
Our project uses techniques of low-temperature geochemistry to interpret and characterize chemical processes, such as precipitation of calcium carbonate in surface lake water (see photos below), occurring in surface water and ground water. We study metal binding by dissolved organic matter and crystal growth and dissolution reaction rate mediation by dissolved constituents because metal ion speciation and mineral formation rates influence water quality and carbon storage. Metal ion complexation with organic matter mediates metal ion reactivity in surface water and ground water -- metal ion speciation controls metal ion chemical and biogeochemical reactivity and bioavailability.
We determine equilibrium metal species distributions in surface and ground water containing organic matter, and characterize crystal growth and dissolution rates in surface and ground water containing organic material.
Project Interests
- Calcium carbonate nucleation and growth in surface and groundwater
- Calcium carbonate formation and dissolution in arid soils at the Amargosa Desert Research Site located near Beatty, Nevada.
Pyramid Lake Research






Aqueous Crystal Growth and Dissolution Kinetics of Calcium Carbonate Minerals at the Amarigosa Desert Research Site, Nevada
Below are publications associated with this project.
Specific surface area of a crushed welded tuff before and after aqueous dissolution
Effects of acid rain and sulfur dioxide on marble dissolution
Welded tuff porosity characterization using mercury intrusion, nitrogen and ethylene glycol monoethyl ether sorption and epifluorescence microscopy
Employment of Gibbs-Donnan-based concepts for interpretation of the properties of linear polyelectrolyte solutions
Modeling electrostatic and heterogeneity effects on proton dissociation from humic substances
Description and testing of three moisture sensors for measuring surface wetness on carbonate building stones
Interpretation with a Donnan-based concept of the influence of simple salt concentration on the apparent binding of divalent ions to the polyelectrolytes polystyrenesulfonate and dextran sulfate
Proton and metal ion binding to natural organic polyelectrolytes-I. Studies with synthetic model compounds
Proton and metal ion binding to natural organic polyelectrolytes-II. Preliminary investigation with a peat and a humic acid
Crystal growth of calcite from calcium bicarbonate solutions at constant PCO2 and 25°C: a test of a calcite dissolution model
Below are software products associated with this project.


