The Edible Aquifer
The Edible Aquifer activity is a hands‑on, food‑based model that helps students visualize how groundwater is stored and moves through different types of aquifers. Using layered ingredients to represent rock, sand, soil, and water, students explore concepts like the water table, recharge, confined vs. unconfined aquifers, and how pollution can spread through groundwater systems.
The Edible Aquifer [PDF]
The Edible Aquifer - DOC

The Edible Aquifer
Objective:
Students will learn and model how groundwater moves through aquifers, how contamination spreads, and compare confined vs. unconfined aquifers using an edible model. This activity aligns with Next Generation Science Standards for middle and high school science education.
Next Generation Science Standards (NGSS) Standards:
- DCIs: ESS2.C: The roles of Water in Earth’s Surface Processes, ESS3.A: Natural Resources, ESS3.C: Human Impacts on Earth Systems
- CCCs: Cause and effect, systems and system models, energy and matter
- SEPs:Developing and using models, analyzing and interpreting data, constructing explanations and designing solutions
Key Terms:
- Aquifer - geologic formation that contains enough water to provide usable quantities to wells or springs.
- Water Table - surface below which all of the pore space in an aquifer is filled with water (saturated).
- Confined Aquifer – an aquifer below the land surface that is saturated with water. Layers of impermeable material are both above and below the aquifer, causing it to be under pressure so that when the aquifer is penetrated by a well, the water will rise above the top of the aquifer
- Unconfined Aquifer - an aquifer whose upper water surface (water table) is at atmospheric pressure and can rise and fall.
- Contamination - Pollutants entering groundwater from the surface or other sources.
Recharge - The process by which water moves from surface water (such as precipitation, rivers, or lakes) to replenish an aquifer.
Background
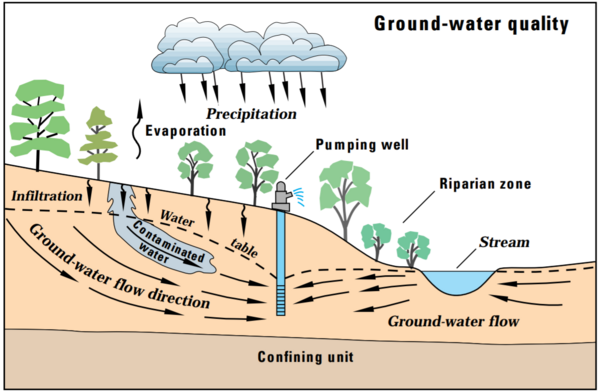
Groundwater is one of the most important sources of drinking water worldwide. It is stored in aquifers. An aquifer consists of layers of rocks and sand beneath the Earth’s surface that hold water in the spaces between particles. Aquifers vary in structure and vulnerability.
An unconfined aquifer has a water table that is open to the surface, meaning water can infiltrate easily from rainfall or surface sources. This makes unconfined aquifers more susceptible to contamination from pollutants like fertilizers, chemicals, and waste.
A confined aquifer, on the other hand, is trapped between impermeable layers such as clay or dense rock. These layers protect the aquifer from surface contamination and cause the water in the aquifer to be under pressure. However, confined aquifers recharge slowly and, if contaminated, are difficult to clean.
Understanding how water moves through these layers and how contamination spreads is critical for managing water resources and protecting drinking water. This activity uses edible materials to model aquifers, allowing students to visualize groundwater movement, the role of different rock or sediment layers, and the impact of pollution.
Building Your Aquifer
Materials
*Choose one material to represent each bolded element. This activity is designed so you can choose ingredients that fit your needs (healthier, allergy‑friendly, or availability).
• Clear cups
• Straw or pipette
• Spoon
• Rocks/gravel - berries, grapes, nuts, chocolate chips, gummy bears, fruit snacks
• Sand – crushed cereal, crushed cookies, crushed graham crackers
• Clay – yogurt, mashed bananas, peanut butter, marshmallow fluff, pudding, ice cream
• Unsaturated zone materials - ice, apples (cut into chunks), freeze dried fruit
• Soil/ land cover – granola, sprinkles
• Groundwater - milk, white grape juice, clear soda, water
• Contamination – food coloring, cocoa powder, kool-aid powder
Alternative, Non-Edible Aquifer: If you prefer a version that doesn’t involve food, you can build a simple aquifer model using the element materials listed above.
Procedure
Each group should built at least build two aquifer models, one confined and one unconfined, to observe differences in water movement, storage, and vulnerability to contamination.
Building Unconfined Aquifer
- Create a layer of rocks/gravel at the bottom of the cup.
Effect: Rocks provide structure and allow water to flow between them, but they don’t store much water themselves. - Add a layer of sand.
Effect: Sand holds water better than gravel, slowing down movement and storing groundwater. - Pour groundwater into the cup until it just covers the previous layers
Observation: Water fills spaces between rocks, gravel, and sand. This is the saturated zone.
• Sand absorbs water more than gravel because of smaller pores.
• In an unconfined aquifer, the top of this saturated zone is called the water table. - Add soil/land cover on top
Effect: This layer interacts with precipitation and can introduce contaminants into the aquifer.
Building Confined Aquifer
- Create a layer of rocks/gravel at the bottom of the cup.
- Add a layer of sand.
- Pour groundwater into the cup until it just covers the previous layers.
- Add a thick layer of clay.
Represents: Confining layer – like clay or dense rock, which is less permeable.
Effect: This layer prevents water from easily passing through and protects the aquifer from surface contamination. - Add unsaturated zone material above the ice cream.
Represents: Unsaturated zone – soil above the confining layer that does not hold groundwater. - Add soil/land cover on top.
Contamination Simulation
- Distribute contamination on top of both aquifers
Observation: In the unconfined aquifer, contamination seeps down quickly into groundwater.
In the confined aquifer, contamination stays mostly on top because the ice cream layer blocks it. - Insert a straw and pump (suck) water from each aquifer
Represents: A well drawing water from the aquifer.
Observation:
• In the unconfined aquifer, contamination reaches the straw quickly.
• In the confined aquifer, water is cleaner because of the protective (confining) layer. - Pour groundwater over the top of aquifer again
Represents: Recharge entering the system as rainfall or precipitation.
Observation: Watch what happens to the Kool-Aid powder (contamination) after this
step.
• In the unconfined aquifer, the Kool-Aid will dissolve and seep downward quickly, showing how surface pollutants infiltrate groundwater.
• In the confined aquifer, the Kool-Aid mostly stays on top because the ice cream layer acts as an impermeable barrier, demonstrating protection from contamination.
Discussion Questions:
Which aquifer is more vulnerable to contamination? Why?
How does the confining layer change water movement?
What happens to the water table when you pump too much water?
After you poured soda over the aquifer a second time (recharge), how quickly did the unconfined aquifer recharged vs the confined aquifer?
- How does this model relate to real-world groundwater protection?
Helpful Links:
https://www.usgs.gov/water-science-school/science/aquifers-and-groundwater
https://www.usgs.gov/water-science-school/science/groundwater-storage-and-water-cycle
*Adapted from Edible Aquifers, The Groundwater Foundation, https://groundwater.org/wp-content/uploads/2025/06/Front-page-EA.pdf

